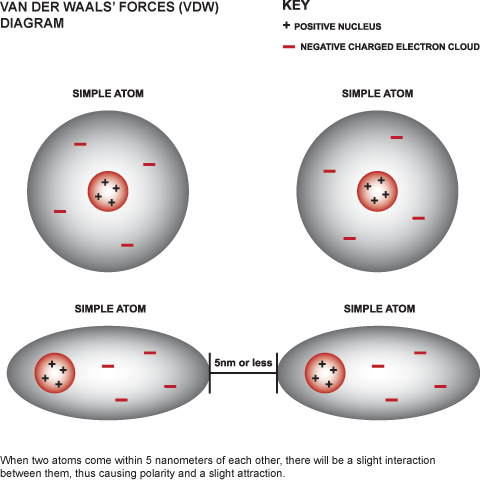
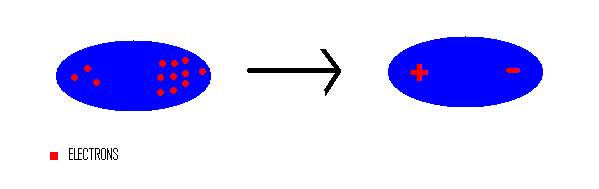
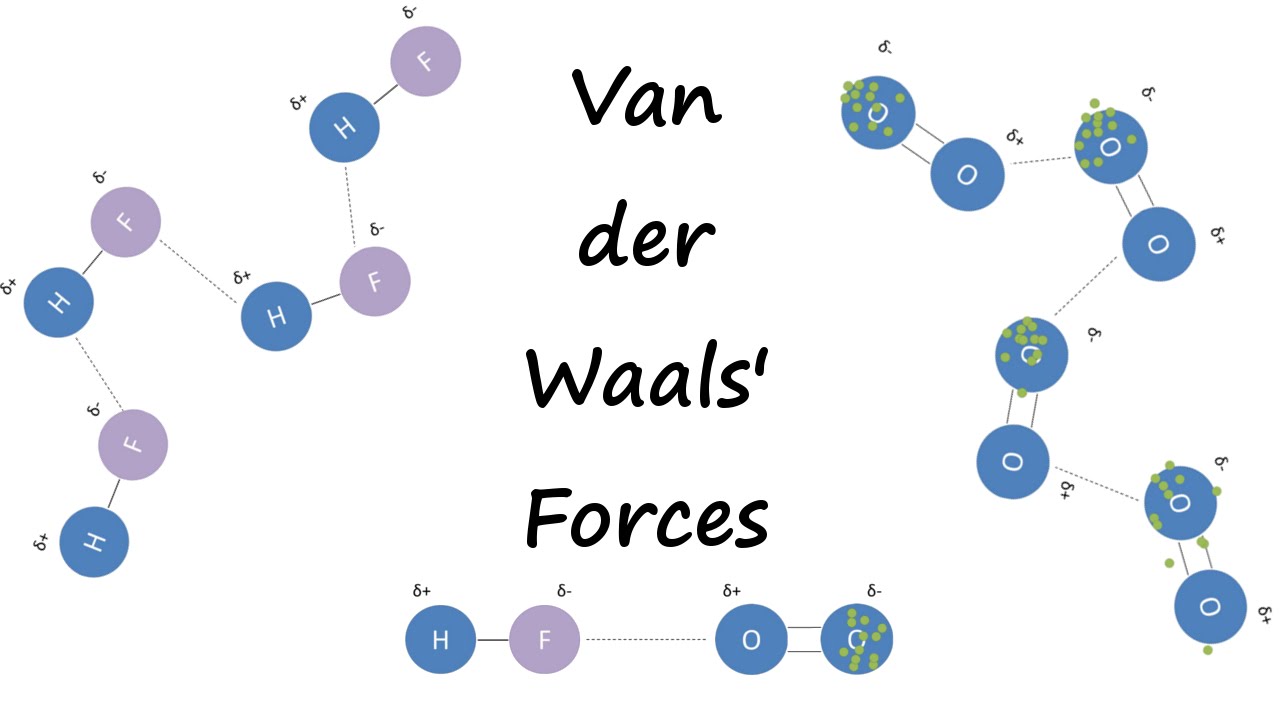
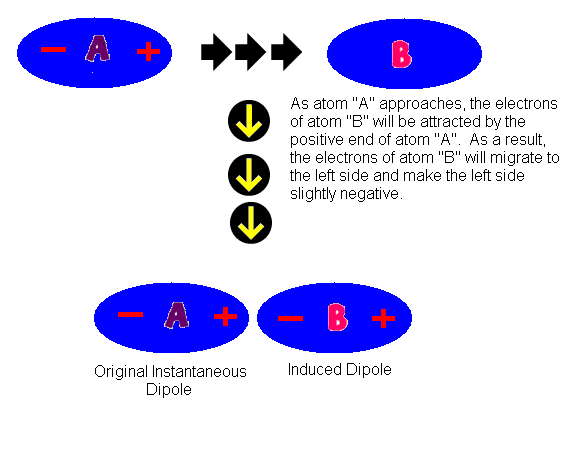
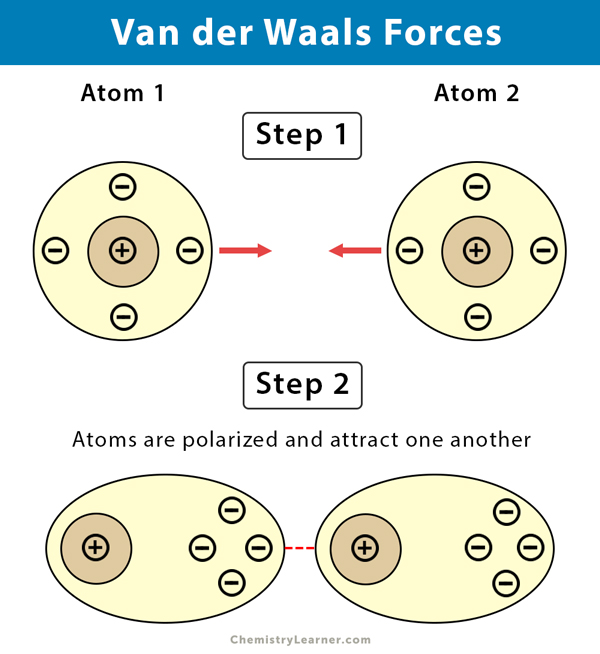
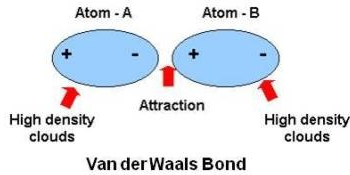
Chemical bond : It is the attractive force that holds two or more atoms together in a molecule or ion. Why do atom Combine? 1. Net attractive force between. - ppt download

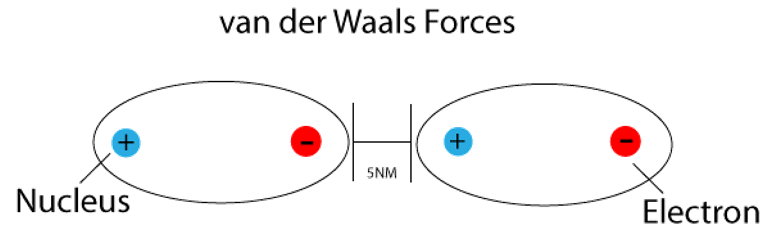
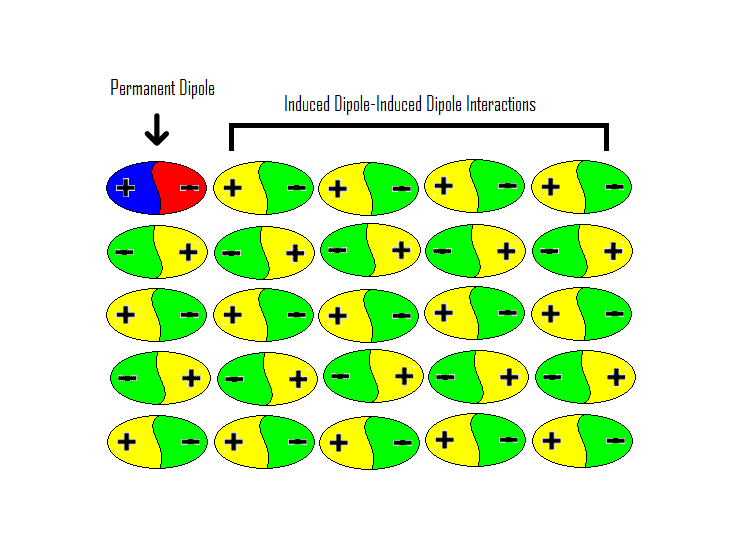
nanoHUB-U FoAFM L1.6: Tip-Surface Interactions (Non-Contact) - InterMolecular Interactions: Dispersion Force
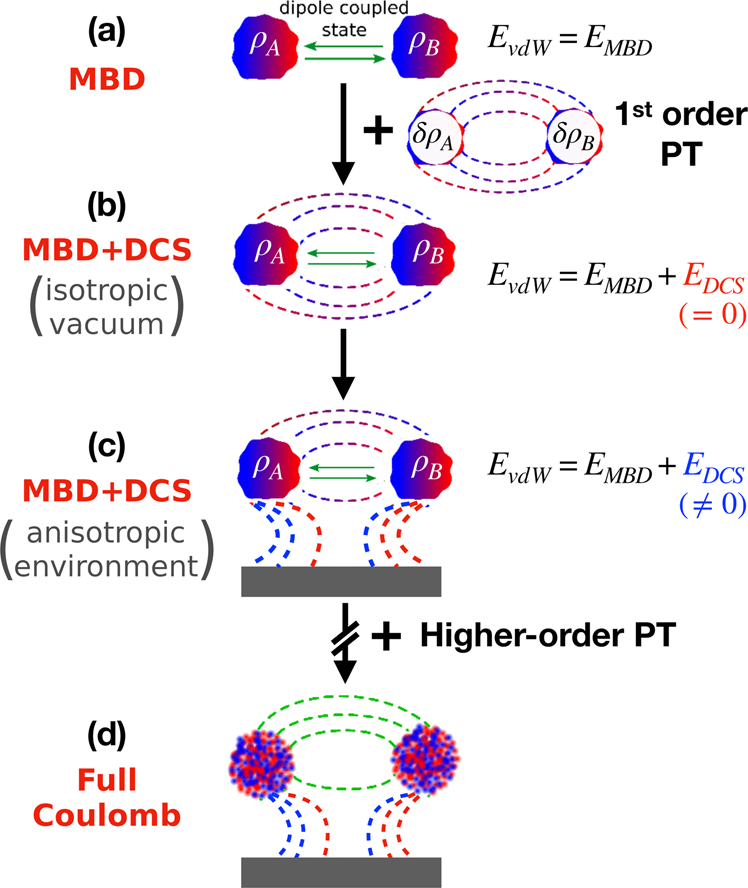
Coulomb interactions between dipolar quantum fluctuations in van der Waals bound molecules and materials | Nature Communications