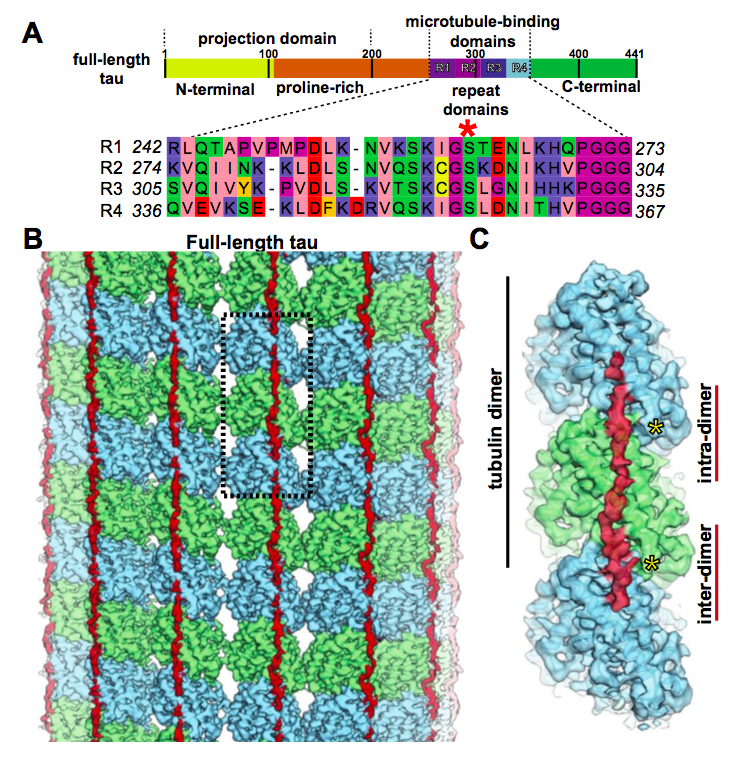
Inability of tau to properly regulate neuronal microtubule dynamics: a loss-of-function mechanism by which tau might mediate neuronal cell death - ScienceDirect
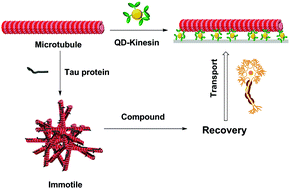
Modulating the microtubule–tau interactions in biomotility systems by altering the chemical environment - Integrative Biology (RSC Publishing)

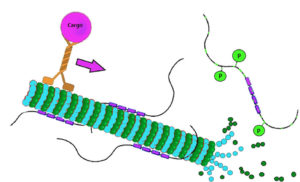
Tau protein. (A) In physiological conditions, tau binds to microtubules... | Download Scientific Diagram
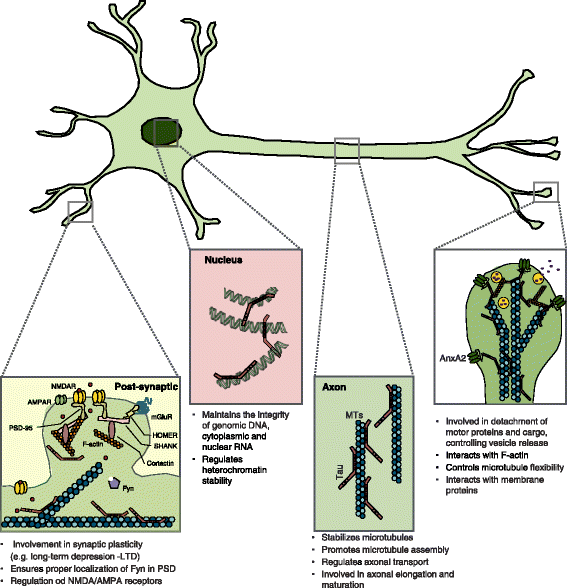
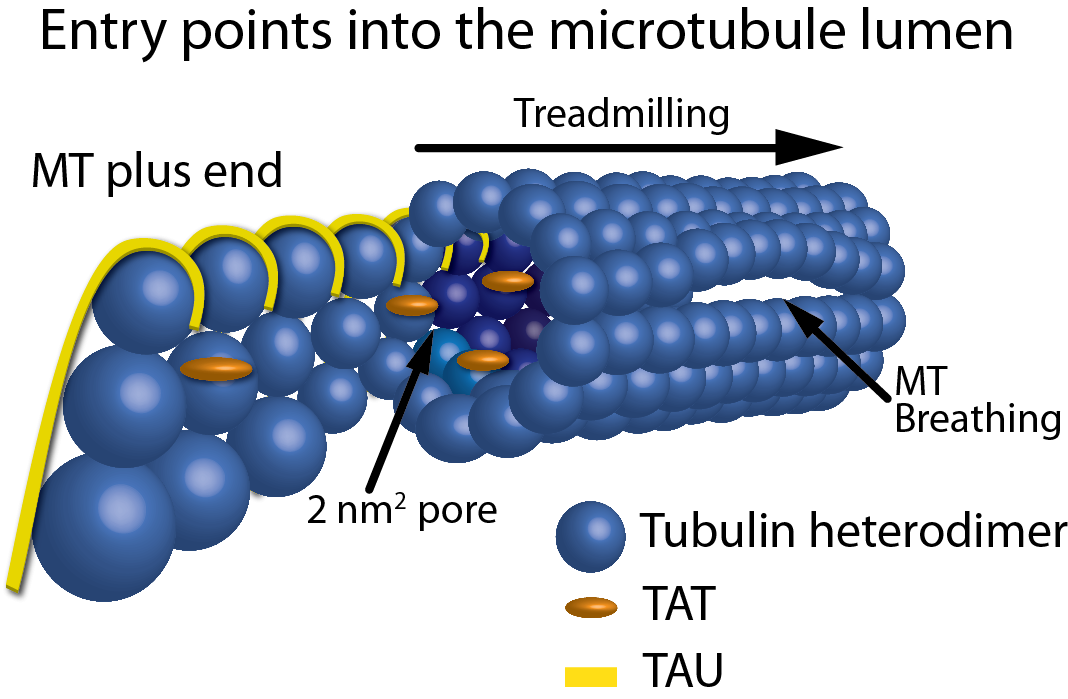
Atypical, non-standard functions of the microtubule associated Tau protein | Acta Neuropathologica Communications | Full Text
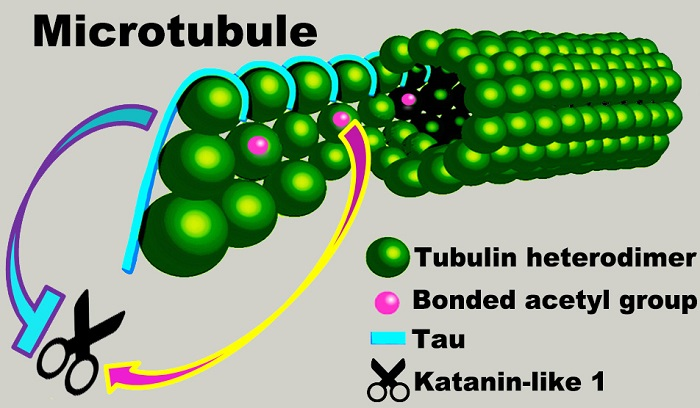
IJMS | Free Full-Text | Microtubule Hyperacetylation Enhances KL1-Dependent Micronucleation under a Tau Deficiency in Mammary Epithelial Cells

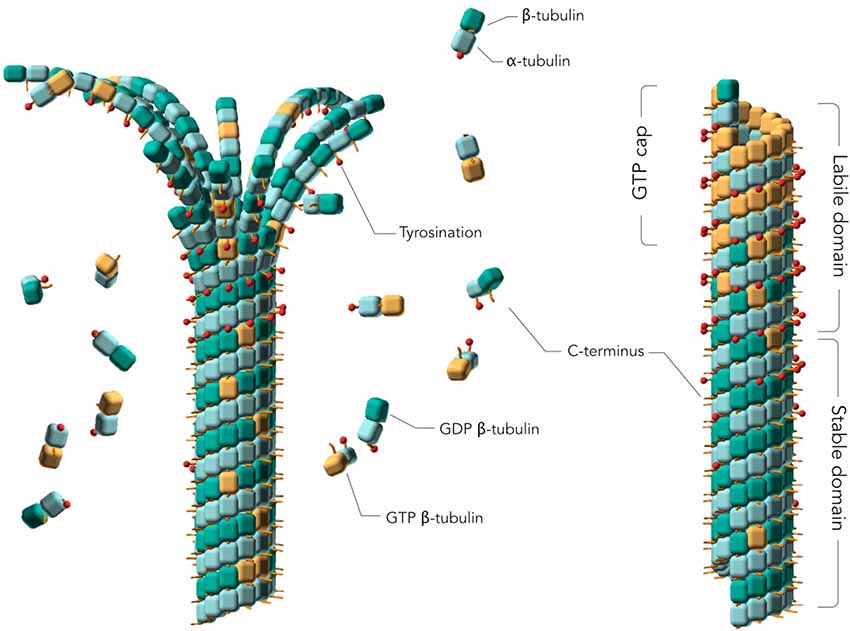
Frontiers | Hyperphosphorylation of Tau Associates With Changes in Its Function Beyond Microtubule Stability
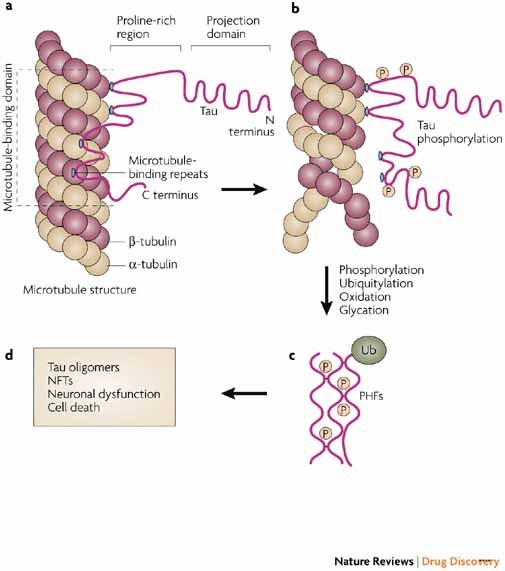
Untangling tau hyperphosphorylation in drug design for neurodegenerative diseases | Nature Reviews Drug Discovery
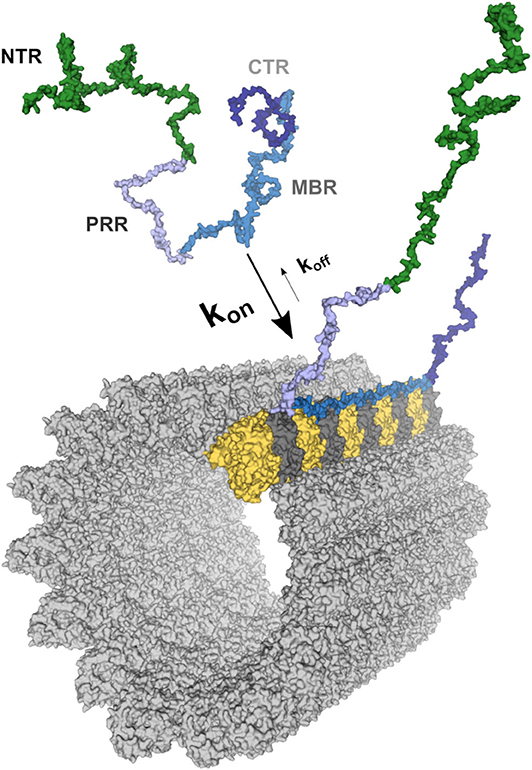
Frontiers | Much More Than a Cytoskeletal Protein: Physiological and Pathological Functions of the Non-microtubule Binding Region of Tau

Minireview - Microtubules and Tubulin Oligomers: Shape Transitions and Assembly by Intrinsically Disordered Protein Tau and Cationic Biomolecules | Langmuir

Oligomerization of the microtubule‐associated protein tau is mediated by its N‐terminal sequences: implications for normal and pathological tau action - Feinstein - 2016 - Journal of Neurochemistry - Wiley Online Library
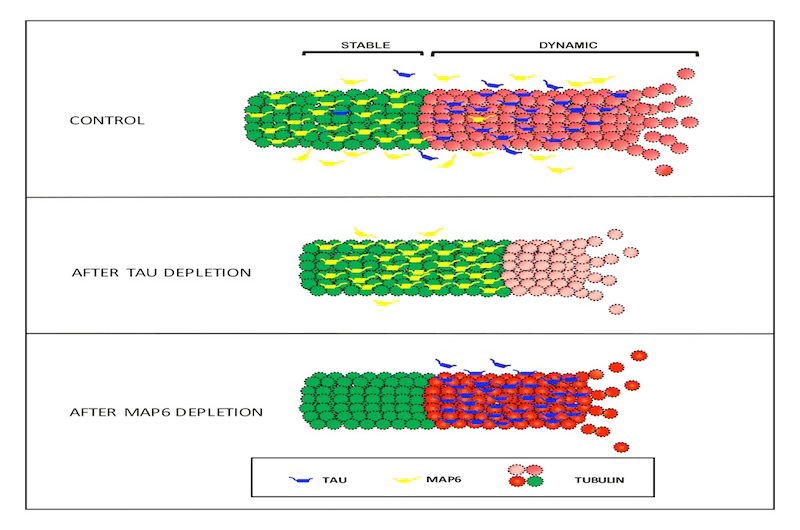
Study Finds Tau Protein Does Not Stabilize Microtubules, Challenges Approach to Treating Alzheimer's

Advances in tau-focused drug discovery for Alzheimer's disease and related tauopathies. - Abstract - Europe PMC

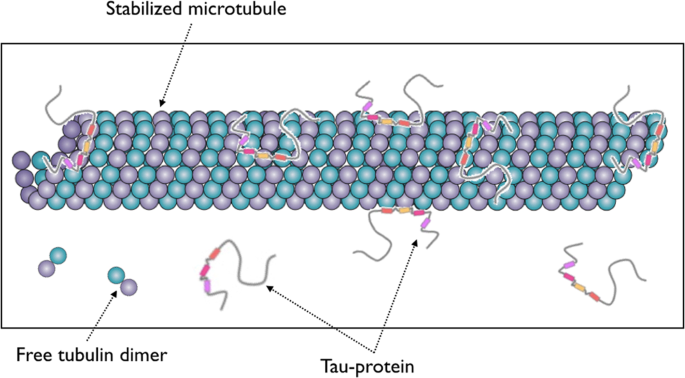
Normal function of tau protein. Tau protein stabilizes microtubules... | Download Scientific Diagram
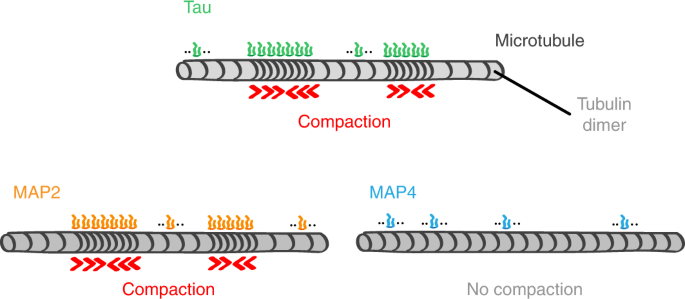



![Microtubule-Associated Protein Tau, Human Recombinant Protein [228-10236] Microtubule-Associated Protein Tau, Human Recombinant Protein [228-10236]](https://www.raybiotech.com/images/thumbnails/280/350/detailed/71/Protein_button.gif)
![PDF] Conformation of Human Microtubule Associated Protein-Tau | Semantic Scholar PDF] Conformation of Human Microtubule Associated Protein-Tau | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/74b85c7c51f3b65a903a99cc0e97e5c642579c9c/8-Figure1.2-1.png)