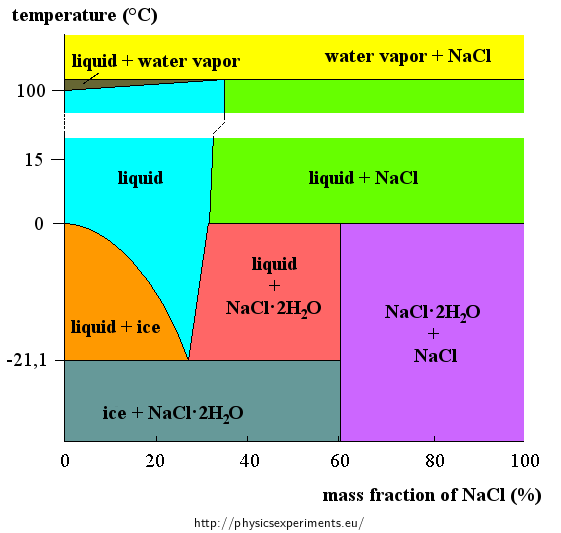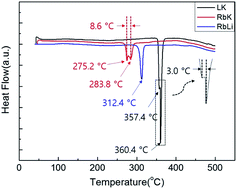
Development of a low-melting-point eutectic salt and evaluation of its discharge performance for light weight thermal batteries - RSC Advances (RSC Publishing)

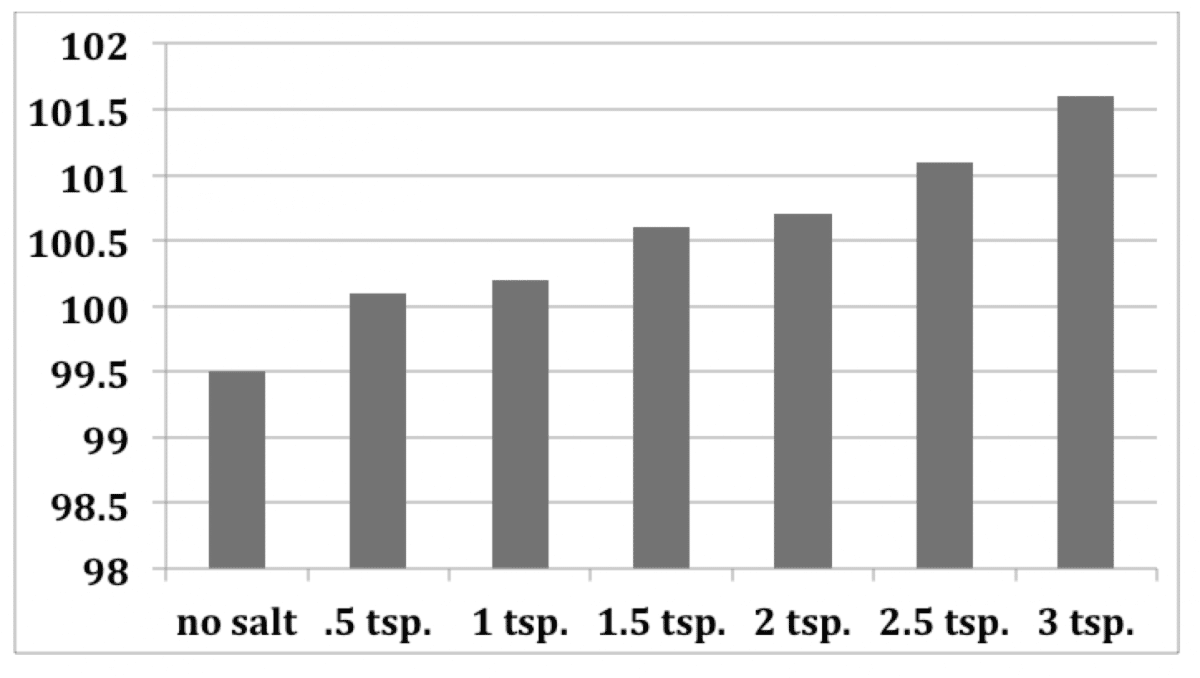
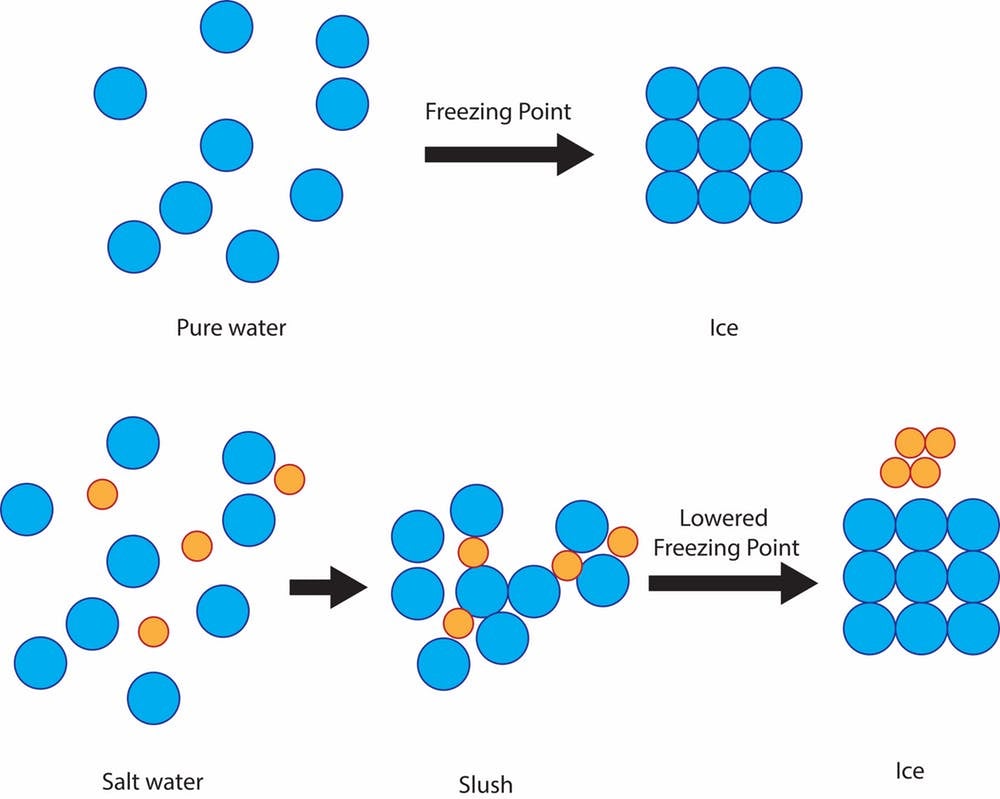
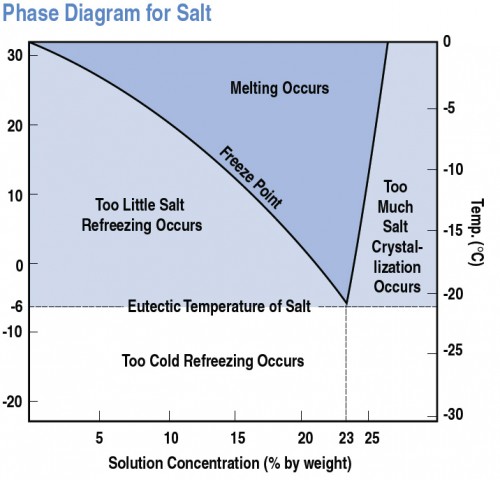
YukonU Youth Moving Mountains - Explore how different substances affect water's freezing point with this fun ice melting experiment! 🧊 Ice forms when water freezes. Pure distilled water has a melting/freezing point