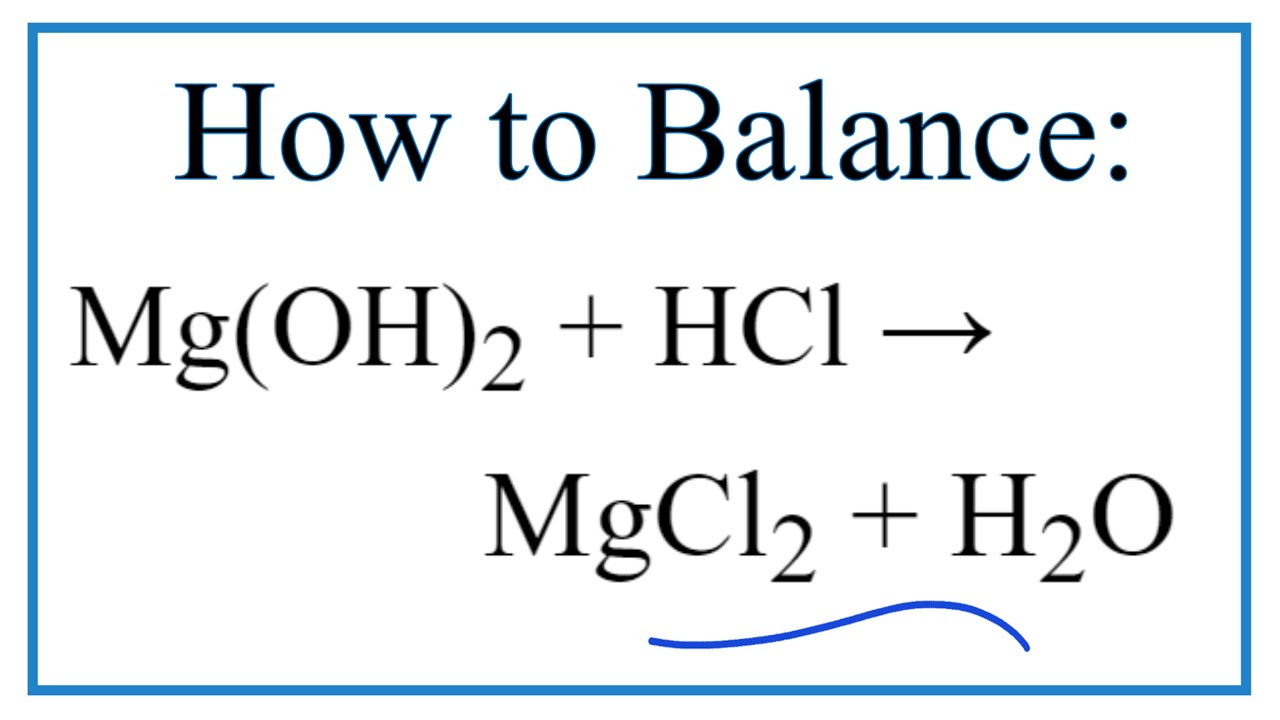
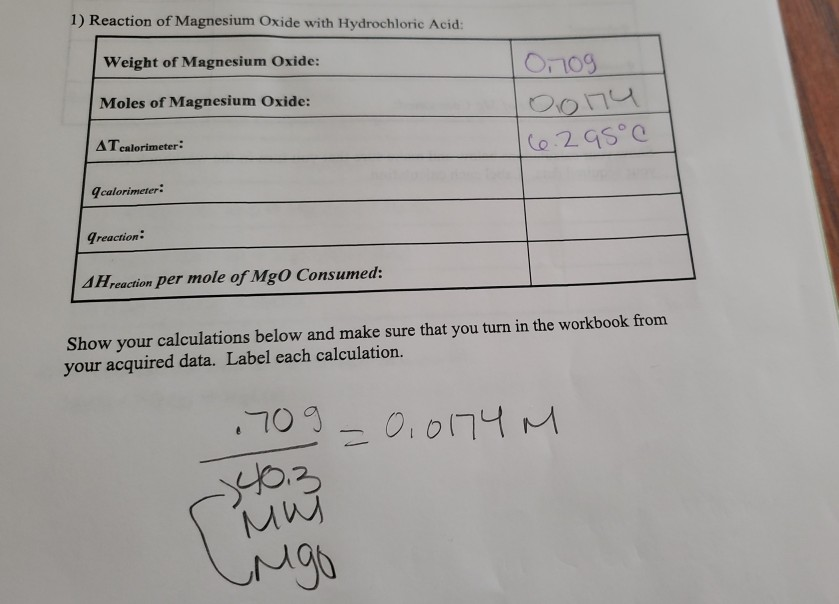
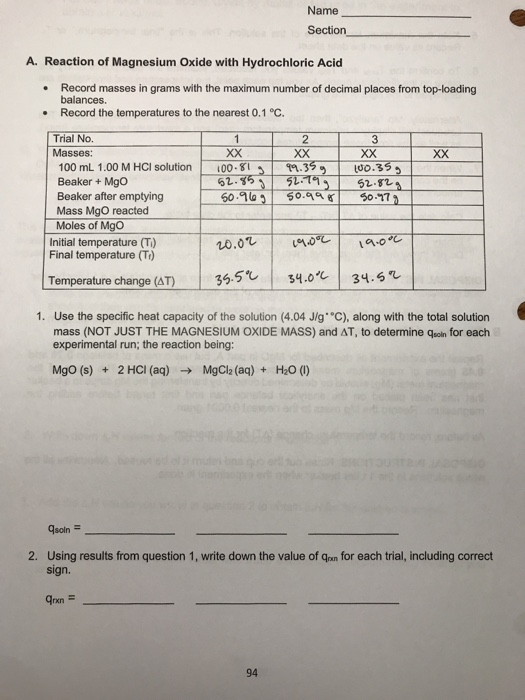
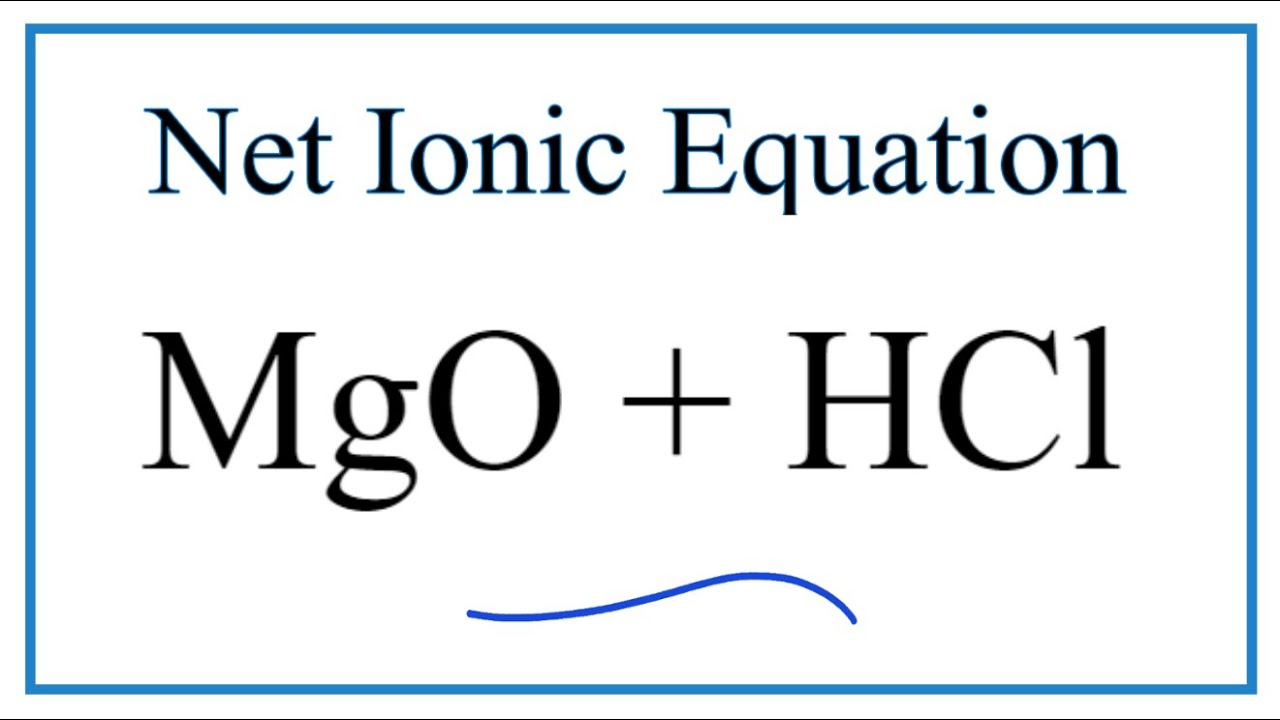SOLVED: 2. Magnesium oxide, MgO, is not very soluble in water, and is difficult to titrate directly. Its purity can be determined by use of a 'back titration' method. 4.06 g of

16/10/2015 Acids and Bases Hydrochloric acidCitric acidWater An acid is a “proton donor”: A base is a “proton acceptor”: H Cl H +- H O Na H O - + Sodium. - ppt download

What is an oxide? An oxide is a Binary compound of oxygen and another element. M & O Oxides can be classified in two ways – Nature of Oxides Amount of. -
Malox, an over-the-counter antacid, contains aluminum hydroxide, Al(OH) 3, and magnesium hydroxides, Mg(OH). What are balanced equations for the reaction of both with stomach acid (HCl)? - Quora