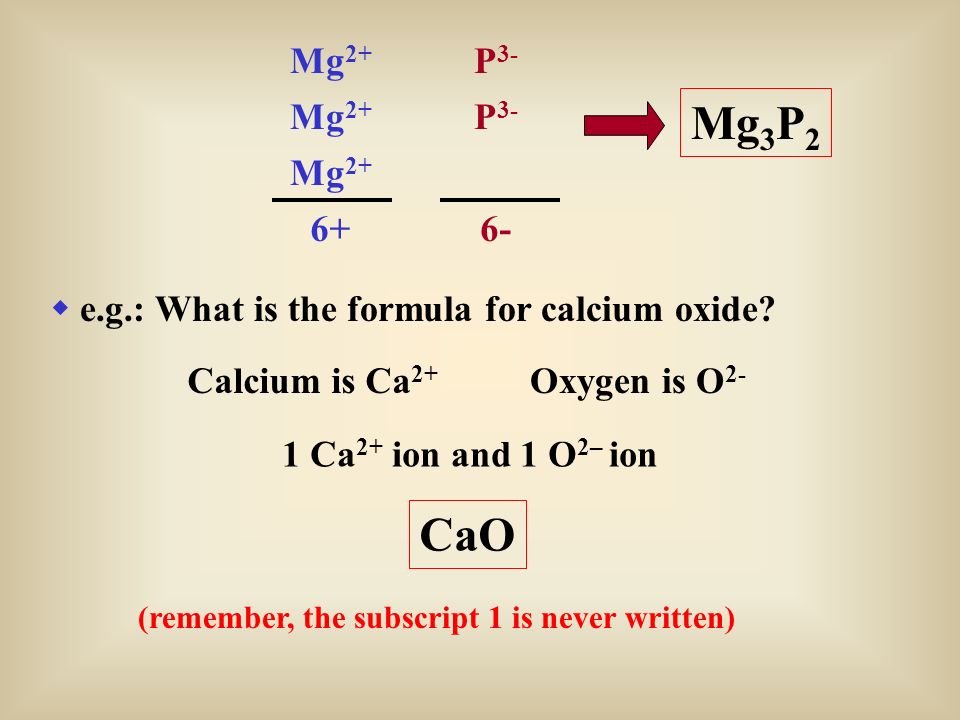
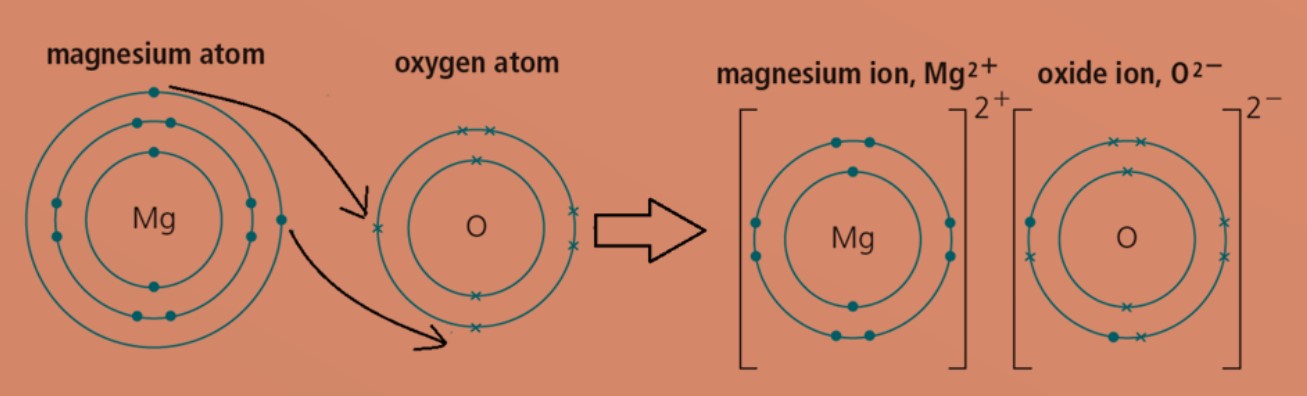
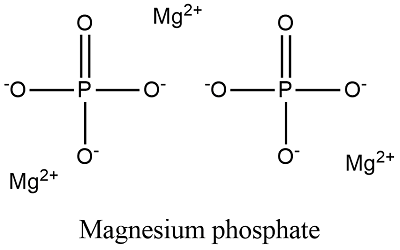
SOLVED: What is the correct formula for an ionic compound that contains magnesium ions and sulfate ions? MgSO4 Mg2SO4 Mg(SO4)3 Mg2S4 MgS4
Magnesium Oxide Formula Mgo Inorganic Compound Occurs Nature Mineral Periclase Stock Video Footage by ©OrangeDeerStudio #465655430