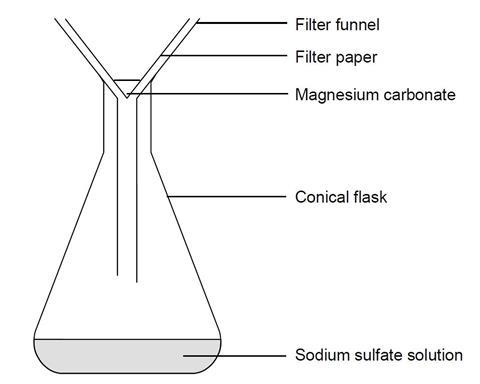
SOLVED: Problem: Carbonic acid (H2CO3) and magnesium carbonate (MgCO3) at certain amounts are added to 1-L de-ionized water, and their initial mole concentrations (t = 0) in DI water were measured as

pH of solutions at various temperatures and pressures with or without... | Download Scientific Diagram
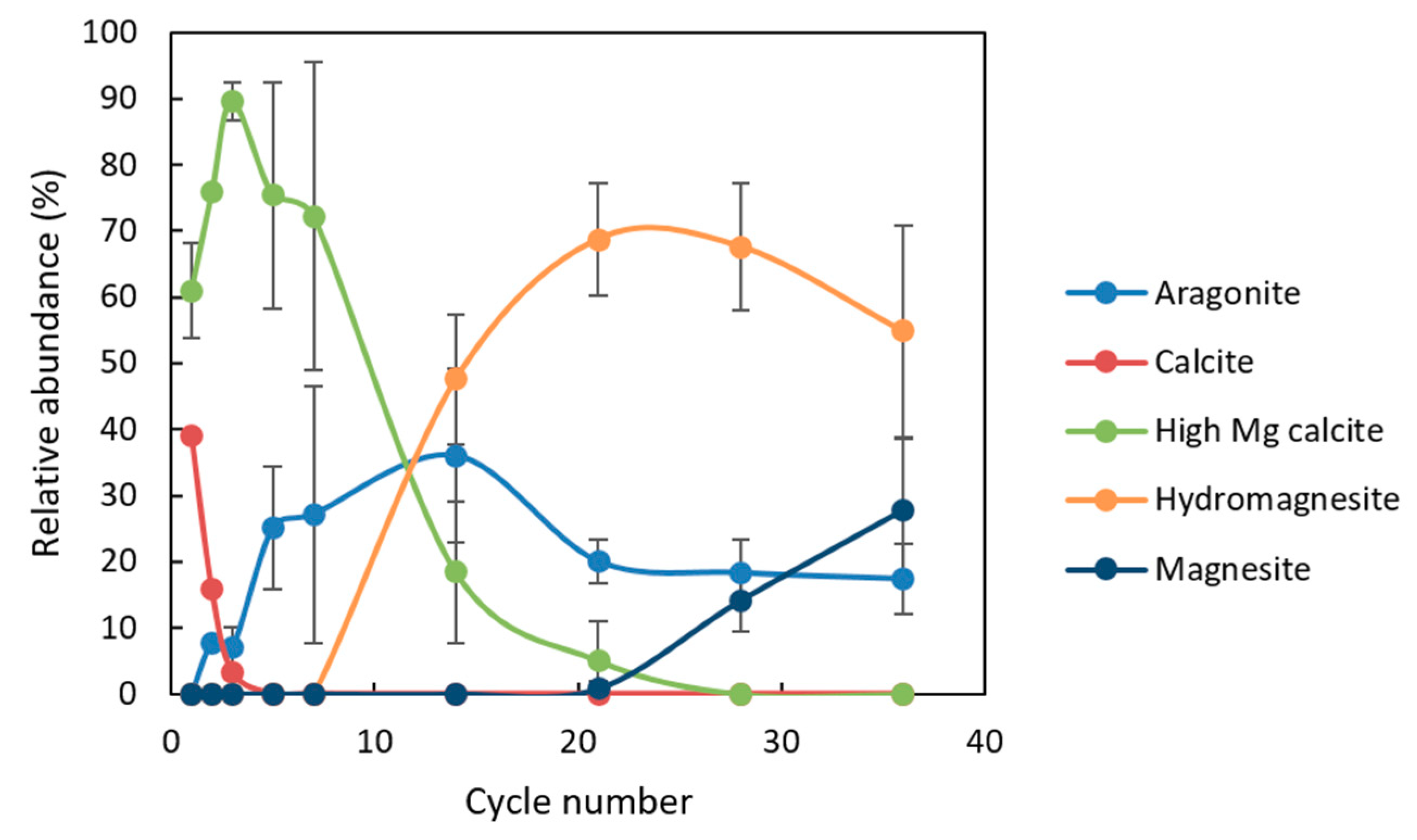
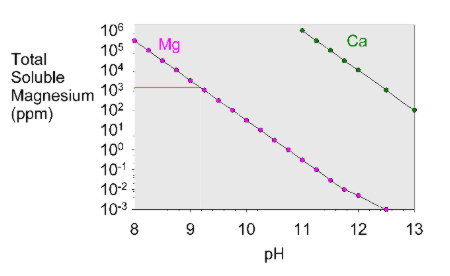
Minerals | Free Full-Text | Effect of pH Cycling and Zinc Ions on Calcium and Magnesium Carbonate Formation in Saline Fluids at Low Temperature
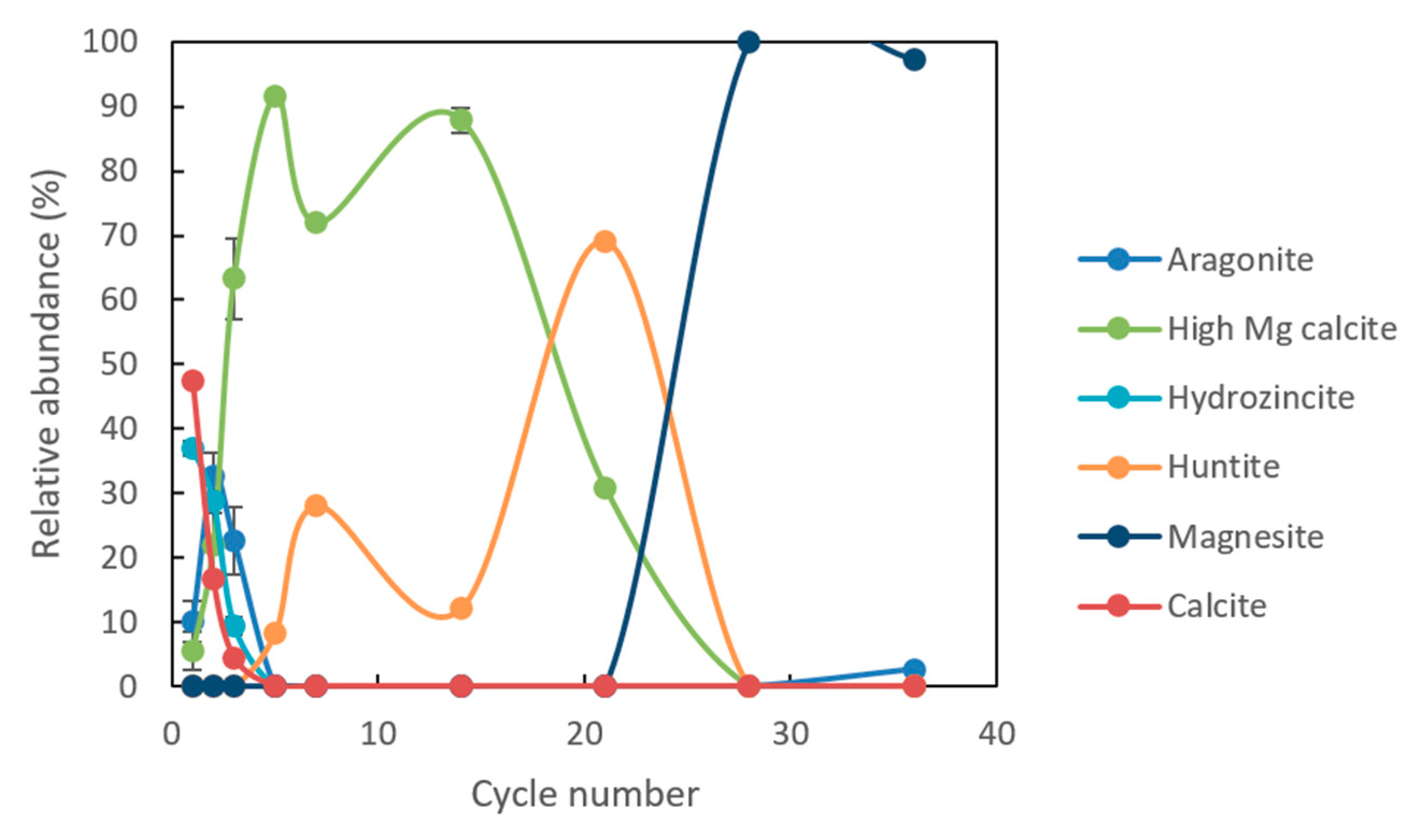
Minerals | Free Full-Text | Effect of pH Cycling and Zinc Ions on Calcium and Magnesium Carbonate Formation in Saline Fluids at Low Temperature

Potentiometric and spectrophotometric study of the stability of magnesium carbonate and bicarbonate ion pairs to 150 °C and aqueous inorganic carbon speciation and magnesite solubility - ScienceDirect
The Kinetics of Magnesium Carbonate Crystallization for Traditional Salt Production Wastewater Recovery

Stability of calcium carbonate and magnesium carbonate in rainwater and nitric acid solutions - ScienceDirect
![PDF] Separation of manganese from calcium and magnesium in sulfate solutions via carbonate precipitation | Semantic Scholar PDF] Separation of manganese from calcium and magnesium in sulfate solutions via carbonate precipitation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0995991619025666d1ab82a34f6fba6a54b8077d/3-Figure1-1.png)
PDF] Separation of manganese from calcium and magnesium in sulfate solutions via carbonate precipitation | Semantic Scholar

Solubility product of amorphous magnesium carbonate - Chang - 2021 - Journal of the Chinese Chemical Society - Wiley Online Library
CO2 Absorption and Magnesium Carbonate Precipitation in MgCl2–NH3–NH4Cl Solutions: Implications for Carbon Capture and Stora

Transformation of abundant magnesium silicate minerals for enhanced CO2 sequestration | Communications Earth & Environment

PDF) Precipitation of MgCO3·3H2O from aqueous solutions: the role of Mg2+ :CO32-concentration ratio, pH and temperature