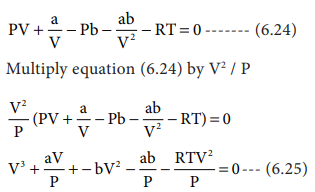SOLVED: Using these equations: a) Derive a formula for the determination of a fugacity using the Van Der Waals equation of state (b) calculate the fugacity of toluene and pyridine at 450K

The equation of state for a van der Waal gas can be expressed as Z = 1 + BVm + CV^2m + ..... If the van der Waal constants a and b
How real gases are different from ideal gases? Derive van der Waal's equation by pressure and volume modifications. - Sarthaks eConnect | Largest Online Education Community

The Derivation of Van Der Waals Equation of State For Real Gases | PDF | Gases | Continuum Mechanics
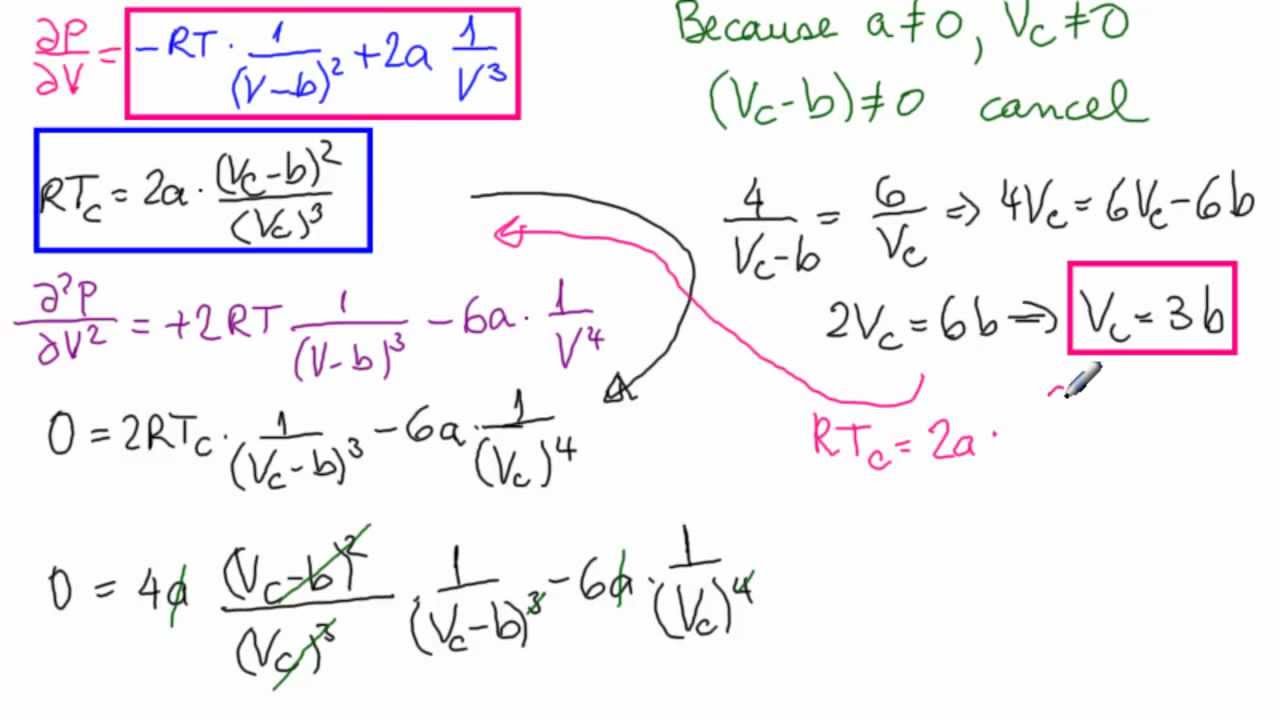
Derive the values of critical constants from the Van der Waals constants. - Sarthaks eConnect | Largest Online Education Community

Reduced form of Van der Waals equation | Law of corresponding states|Unit 5| BPC Class |Dr. M. Ghosh - YouTube

OneClass: here Evaluating a derivative of the van der Waals equation using the cyclic rule Find the p...
![SOLVED: (a) Explain what is meant by the equation of state of a substance. You can assume single phase one-component system [2] The van der Waals equation of state for one mole SOLVED: (a) Explain what is meant by the equation of state of a substance. You can assume single phase one-component system [2] The van der Waals equation of state for one mole](https://cdn.numerade.com/ask_images/0e8a1e7ae3f94d79a8a4701a0de80b7e.jpg)