The density of water at 4 degree celsius is 1 gram per centimetre cube find its value in s i unit - Brainly.in

The density of water at `4^(@)C` is `1.0 xx 10^(3)kg m^(-3)`. The volume occupied by one molecule of - YouTube

The density of water at 4^(@)C is 1.0 xx 10^(3)kg m^(-3). The volume occupied by one molecule of water is approximately (N(A) = 6.0 xx 10^(23)):

The density of water at 4^oC is 1.0 × 10^3 kg m^-3 . the volume occupied by one molecule of water is approximately:

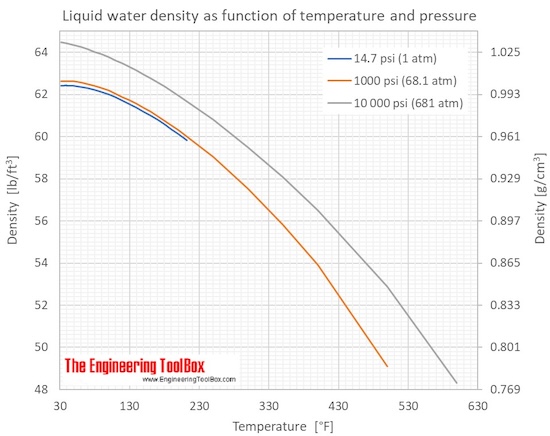
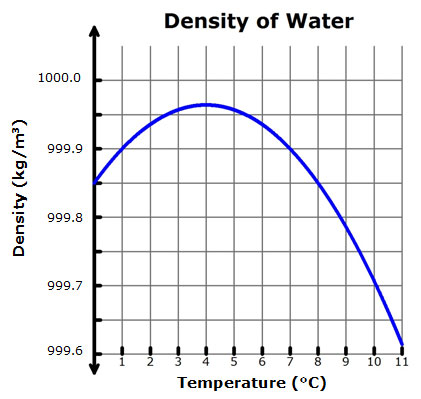
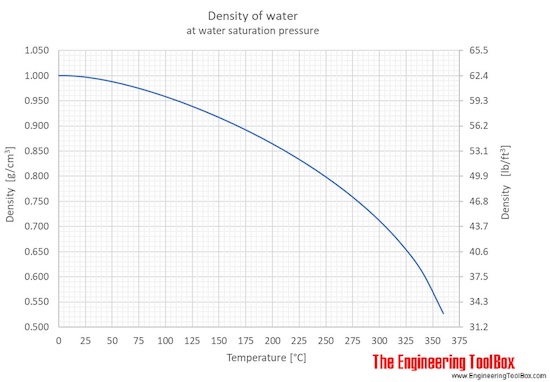
Using data from the figure, graph the density as a function of the temperature for liquid water from 100°C to 4°C. | Wyzant Ask An Expert

Determination of the Density of Water at 4° C. by the International Bureau of Weights and Measures, 1899 | Science