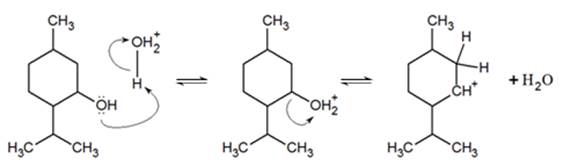
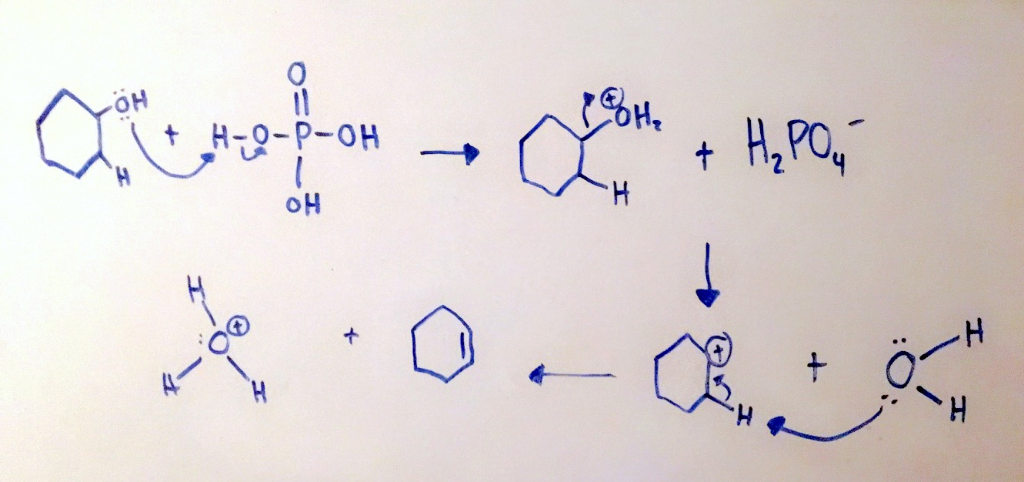
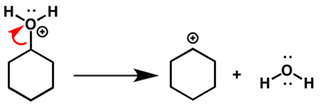
Predict the major product of acid catalysed dehydration:(i) 1 - methyl cyclohexanol(ii) Butan - 1 - ol

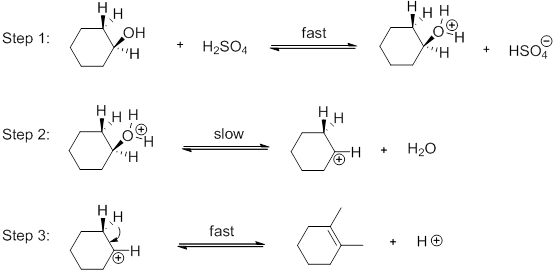
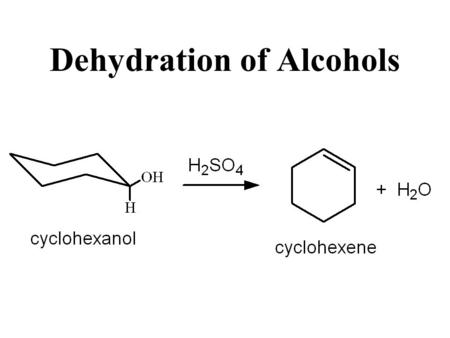
The reactions shown above illustrate the mechanism of the dehydration process. They are all shown as reversible processes. a.) Explain why the synthes is process of cyclohexene from cyclohexanol perm | Homework.Study.com

Write out a detailed mechanism of the acid catalyzed dehydration of cyclohexanol. use only hydronium - Brainly.com
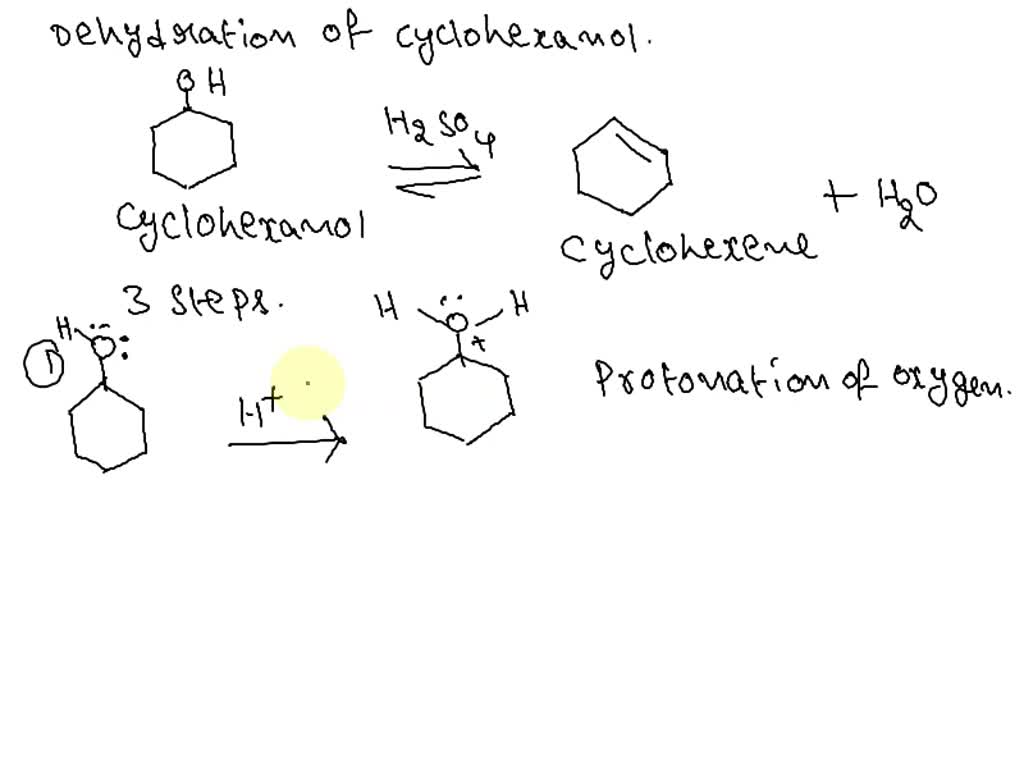
SOLVED: Write out a detailed mechanism of the acid catalyzed dehydration of cyclohexanol. Use only hydronium as your acid source.

Why the concentrated hydrochloric acid be a poor choice as the acid catalyst for formation of cyclohexene by dehydration of cyclohexanol? | Homework.Study.com
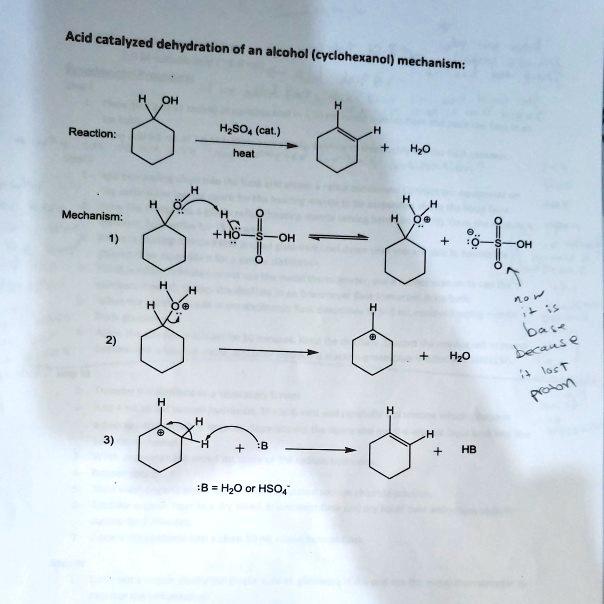
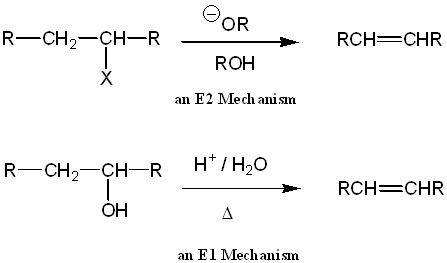
SOLVED: Acid catalyzed dehydration of an alcohol (cyclohexanol) = mechanism: Reactlon: HzSO (cat ) heat Hzo Mechanism; HzO 'B = HzO or HSO4" 0v 3C4 - 0 lec T Rol ^