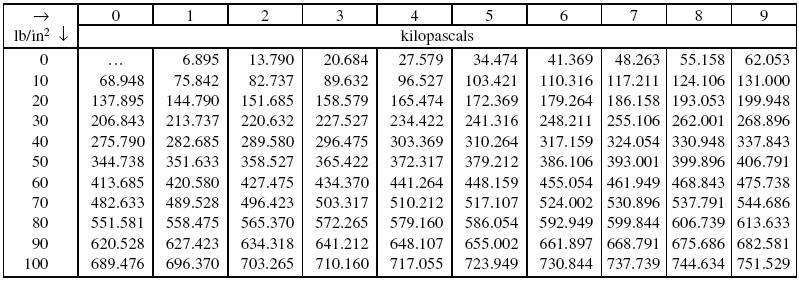
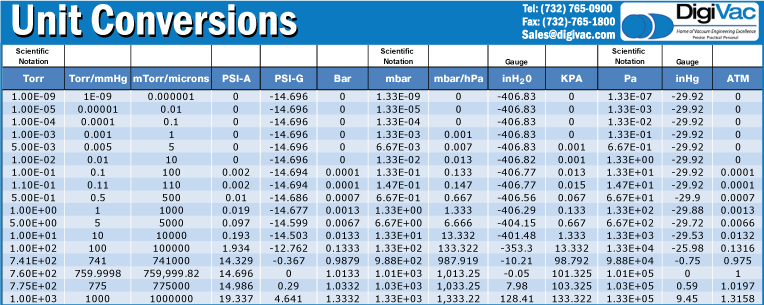
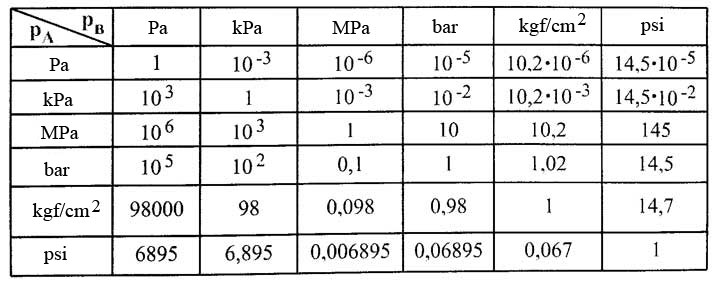
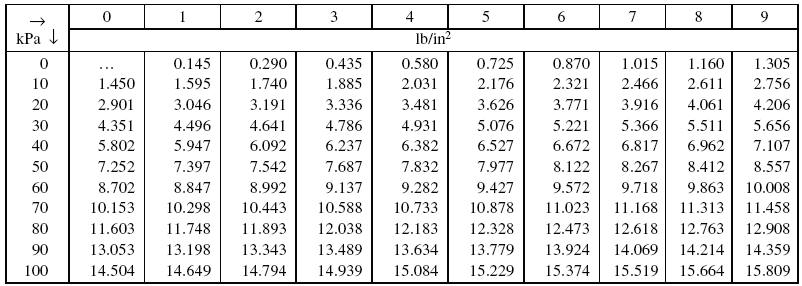
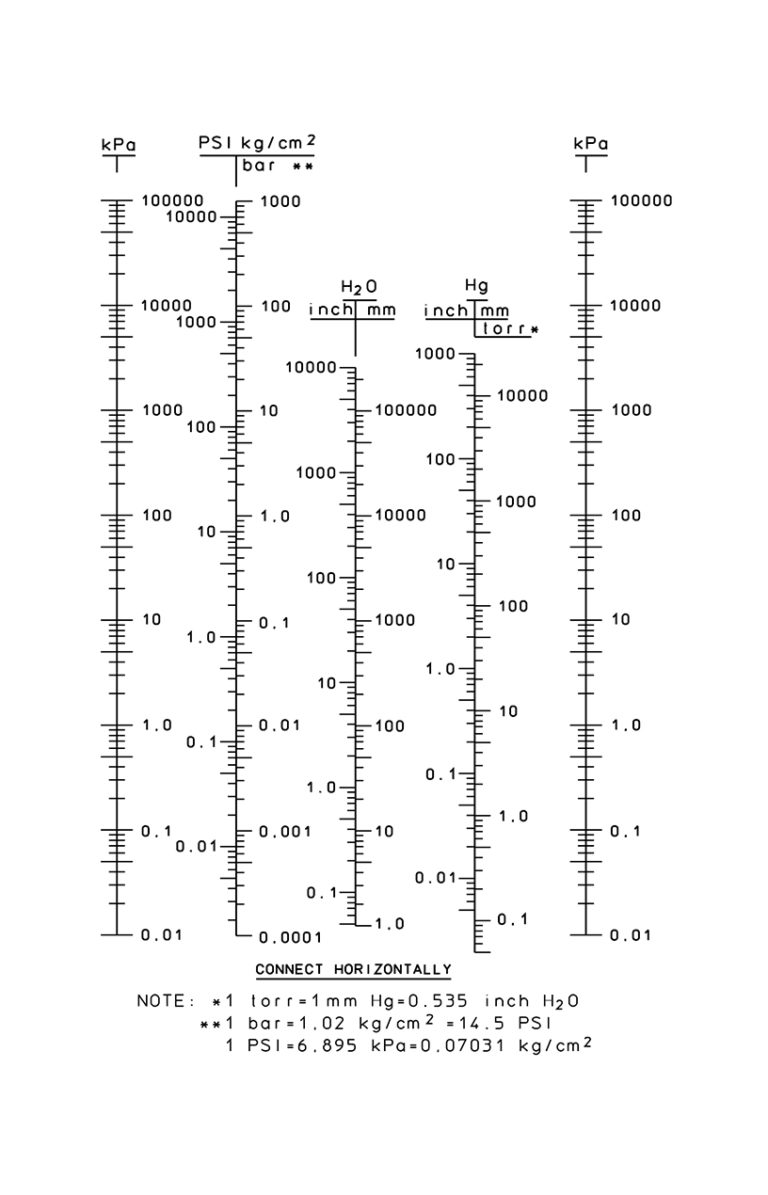
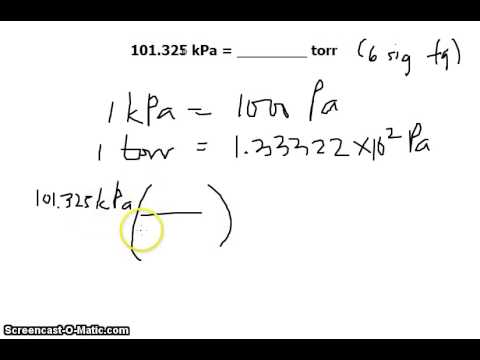SOLVED: Convert the following pressures. 109.2 kPa 822 torr 9.75 psi 764 mmHg 697 torr alm mm Hg mm Hg kPa kPa Pretend that you're talking to a friend who has not
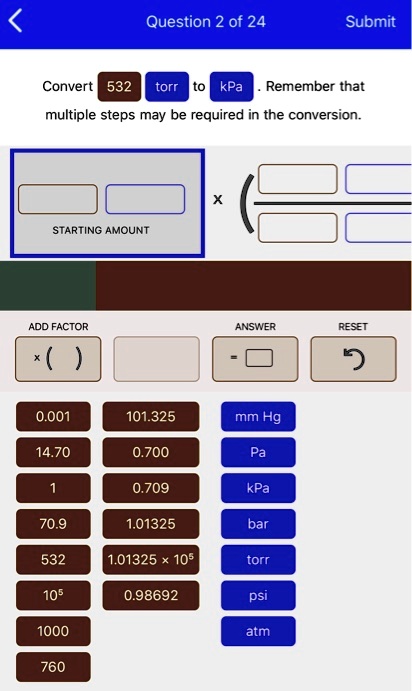
SOLVED: Question 2 of 24 Submit Convert 532 torr kPa Remember that multiple steps may be required in the conversion: STARTING AMOUNT ADD FACTOR ANSWER RESET 0.001 101.325 mm Hg 14.70 0.700 0.709 kPa 70.9 1.01325 bar 532 1.01325 105 torr 105 0.98692 psi ...
![B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book] B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book]](https://www.oreilly.com/api/v2/epubs/9780134663906/files/graphics/f0755-01.jpg)
B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book]