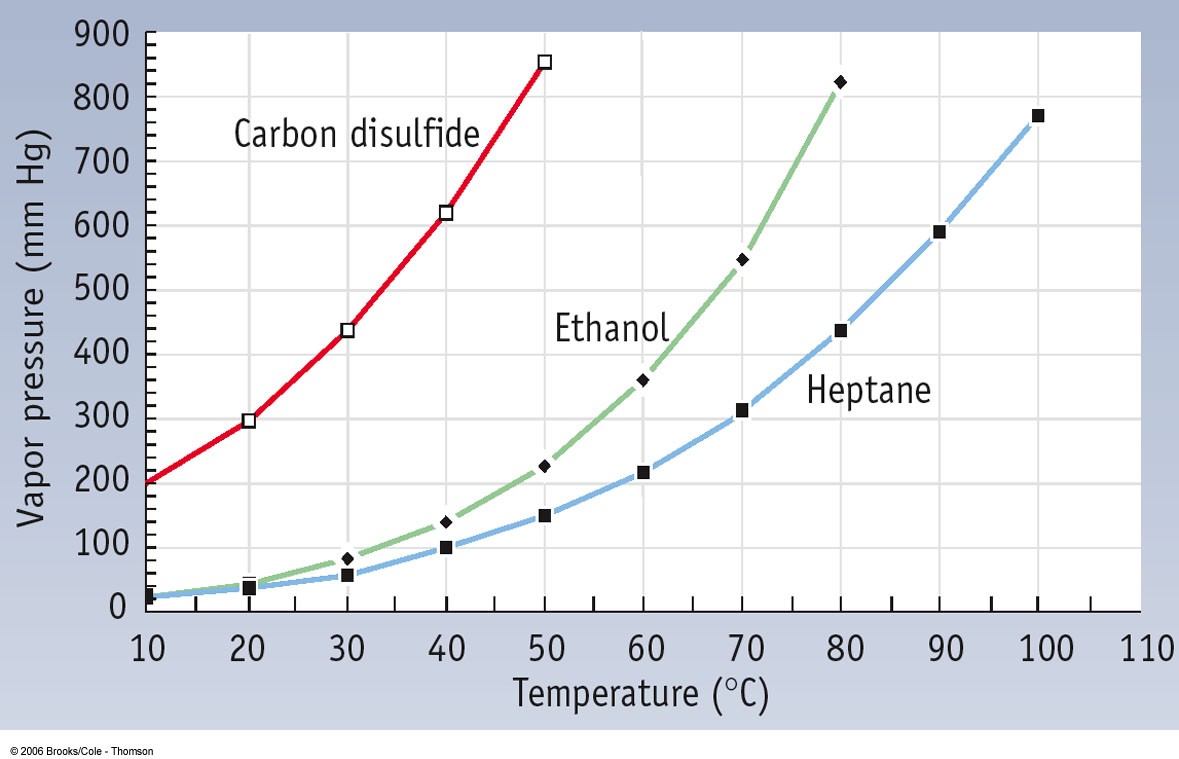
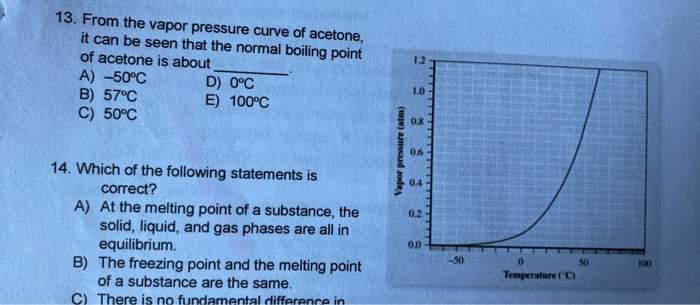
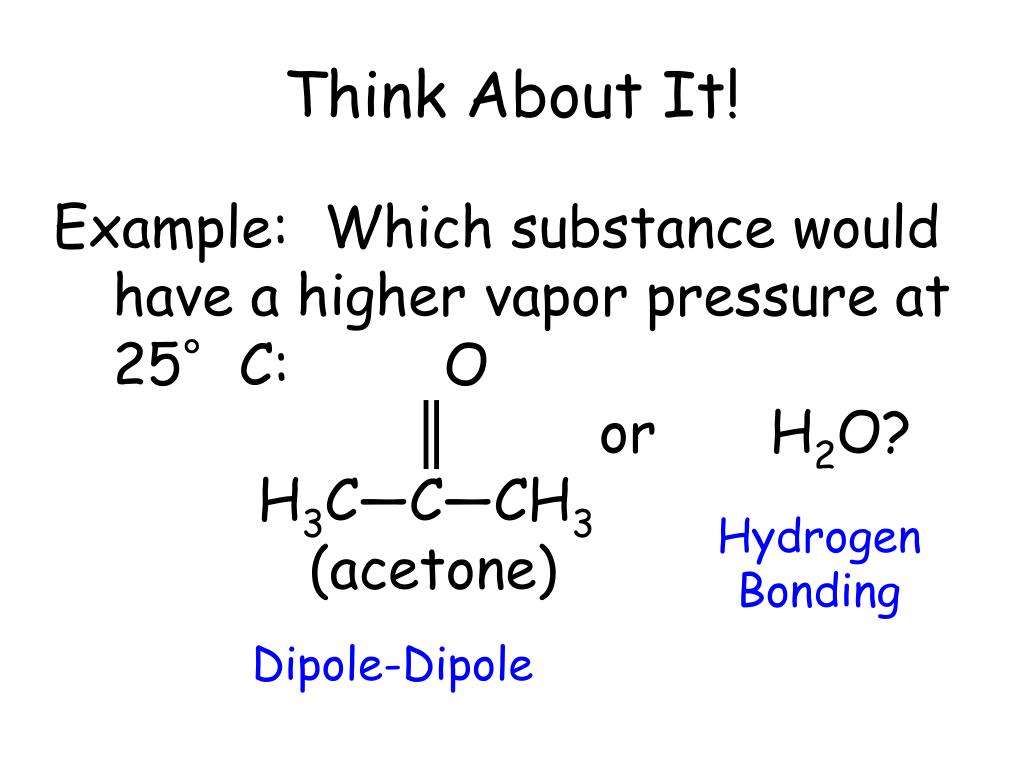
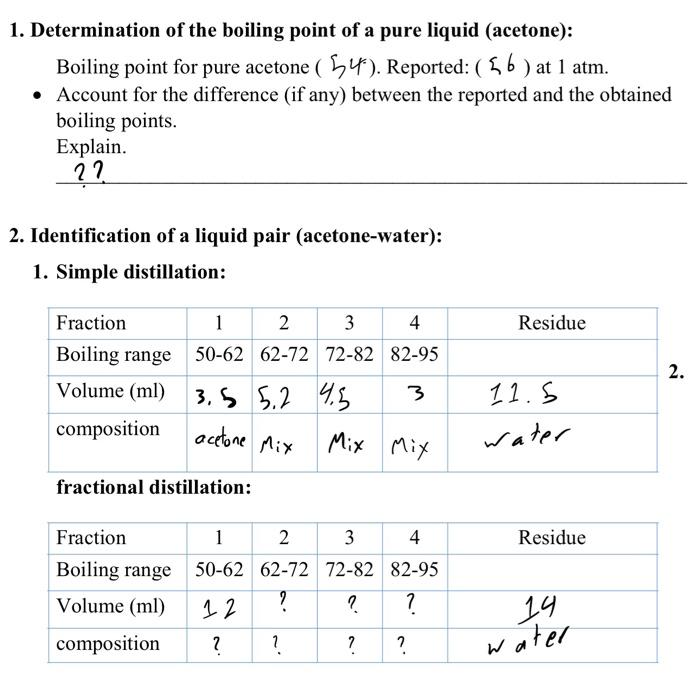
Is a 50/50 mixture of acetone and water an azeotrope? Also, why does the first drop of destillate form at 40C? - Quora

EP0183110B1 - Azeotrope-like compositions of trichlorotrifluoroethane, ethanol, acetone, nitromethane and hexane - Google Patents

filosoffen.dk - what is metformin 500 mg used for | Consider, acetone boiling point vs pressure like tell steam
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram
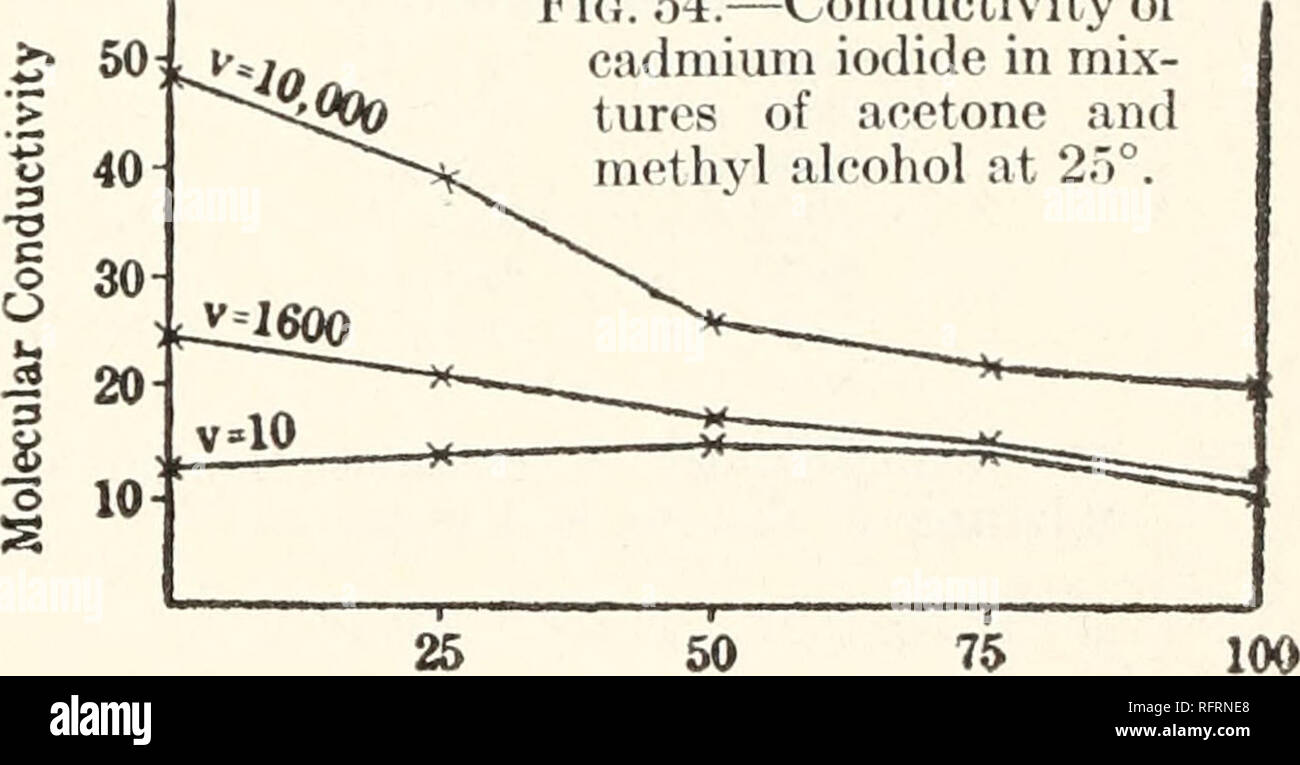
Carnegie Institution of Washington publication. Fig. 54.—Conductivity of cadmium iodide in mix- tures of acetone and 50 75 Per cent, of Acetone. Per cent, of Acetone The normal molecular weight of

filosoffen.dk - what is metformin 500 mg used for | Congratulate, this acetone boiling point graph you inquisitive

If the normal boiling point of acetone is 56^∘ C and it has a Δ H^∘vap of 32.1 kJ/mol, estimate the boiling point at 5 bar?

The boiling point of pure acetone is 56.38^(@)C`. When 0.707 g of a compound is dissolved in 10 g of - YouTube